AdvancellsTM Molecular Transport Medium
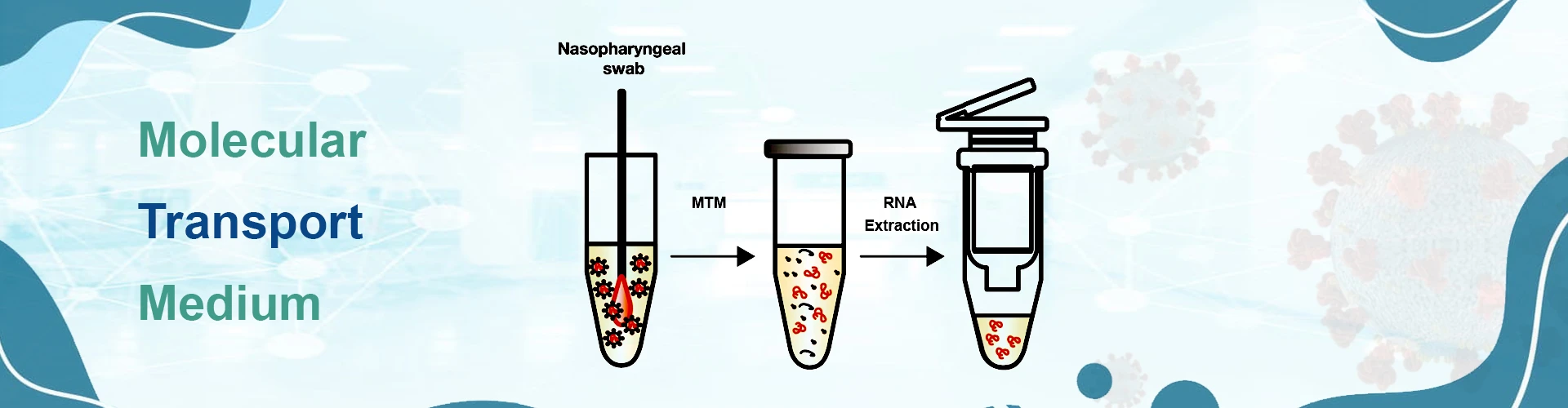
Another variant of AdvancellsTM COVID 19 transport solutions, and is specifically designed with an intent to safely transport intact nucleic acid material from pathogenic viruses, such as but not limited to Sars-CoV-19 from the collection centres to the diagnostic testing centres. As per the latest guidelines proposed by CDC and WHO, it is important to promptly respond to the current pandemic situation with the help of accurate testing, immediate containment and offering appropriate treatment facilities. Accordingly, the ICMR has recommended molecular testing of the virus nucleic acid material with the help of quantitative real time PCR method (qRT-PCR).
The molecular transport medium is a unique formulation designed with the help of guanidine Thiocynate an appropriate chelating agent that destroys protective coating of the virus membrane and extract inside molecular machinery. The step is very critical and needs to be followed in every transport system, whether VTM, VLM or MTM. Through molecular transport medium i.e. MTM we are presenting a very robust system to extract intact RNA from clinically positive specimen, stabilize the same in appropriate buffering formulation; and is further helpful in extracting intact nucleic acid material at the diagnostic centres. Designed and optimized for all types of molecular applications, including qRT-PCR and next generation sequencing; the AdvancellsTM Molecular Transport Medium provides a very robust system for nucleic acid extraction

Key Features of MTM Kits
- 1.5 ml Molecular Transport Medium in 5 ml ETO sterilized flat base tube
- Unique formulations are helpful for viral inactivation immediately, yet maintaining the molecular integrity of the virus.
- Flocked nylon swab with higher surface area for effective capture of mucosal epithelial cells infected with Virus.
- Enhanced stability up to 24 months and accurate diagnosis even at low viral load.
- Since the system is comprised of inactivated viral particles, it offers complete protection to the end-users.
- Each lot is QC tested for fungal/microbial growth and chemical parameters
- The batch is released with the appropriate Certificate of Authenticity
- The product is validated by ICMR/NIV for optimum performance.

Detail Instruction for use
- Open the swab first, check for the sterility performance of the swab by checking its indicator.
- Place the swab appropriately in the buccal cavity, by asking patients to open their mouths wide apart.
- Rub the swab gently against the inner lining of the mucosa (The pinkish part of the pellet in the mouth)
- Beware to protect yourself with appropriate protective wears like gowns, masks, eye shields, gloves, etc.
- Cut the plastic shaft at the breaking point and place it in the appropriate tube; tighten the cap, and seal it tightly.

Quality Control
Advancells Diagnostics MTM kits are manufactured in sterile conditions and meet the ISO Guidelines.

Appearance
Transparent Clear solution

pH at 25°C
7.4 ± 0.2


Osmolality in mOsm/Kg H2O
270mOsm - 290mOsm

Sterility
No bacterial or fungal growth was observed after 14 days of incubation.